Pfizer submits to FDA for approval of 3 doses of COVID-19 vaccine for children aged 5-11
Pfizer and BioNTech said that they have applied to the US Food and Drug Administration (FDA) for an emergency authorization to use a booster dose of COVID-19 vaccine for children aged 5-11 years.
This pharmaceutical company said that a 3-dose dose of COVID-19 vaccine aimed to increase anti-Omicron antibodies 36 times for ages 5-11.
In an analysis of 140 children who had never had COVID-19 before, antibodies against the original strain of SARS-CoV-2 were six times higher 1 month after dose 3 compared with dose 2.
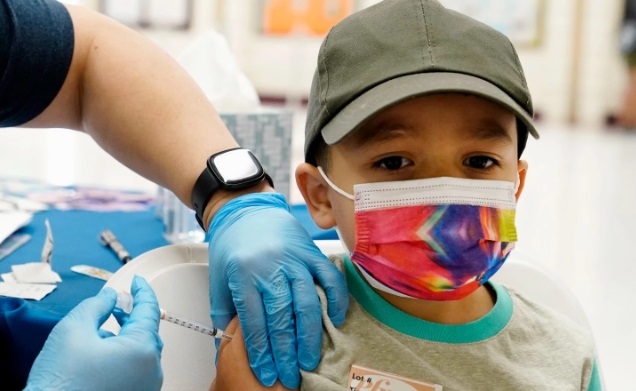
Pfizer said that giving three doses of COVID-19 vaccine to children aged 5-11 years increased the amount of Omicron antibodies 36 times.
Research from the New York State Department of Health and the US Centers for Disease Control and Prevention (CDC) shows that the anti-infective effectiveness of the Pfizer vaccine for children aged 5-12 is significantly reduced due to the Omicron wave, reduced from 68% to about 12% of the anti-infective effect.
However, getting two full doses of the COVID-19 vaccine continues to protect children from severe COVID-19 disease. This figure is evident in hospitalizations or emergencies due to COVID-19.
More than 10,000 children aged 6 months to 12 years have participated in a worldwide trial of Pfizer’s COVID-19 vaccine.
Pfizer hopes to submit data on trials in children under the age of 5 in the coming months.
Children under 5 are the only age group that has not yet been approved for a COVID-19 vaccine in the United States.
Pfizer/BioNTech said it plans to submit the data to the European Medicines Agency and other regulators for approval in the coming weeks.
Pfizer’s two-dose course of COVID-19 vaccine for children aged 5-11 was approved in the US last October.
Booster doses are also available for immunocompromised children 12 years of age and older, as well as for adults. A four-dose dose of the COVID-19 vaccine has been approved for anyone 50 years of age or older.
at Blogtuan.info – Source: Afamily.vn – Read the original article here